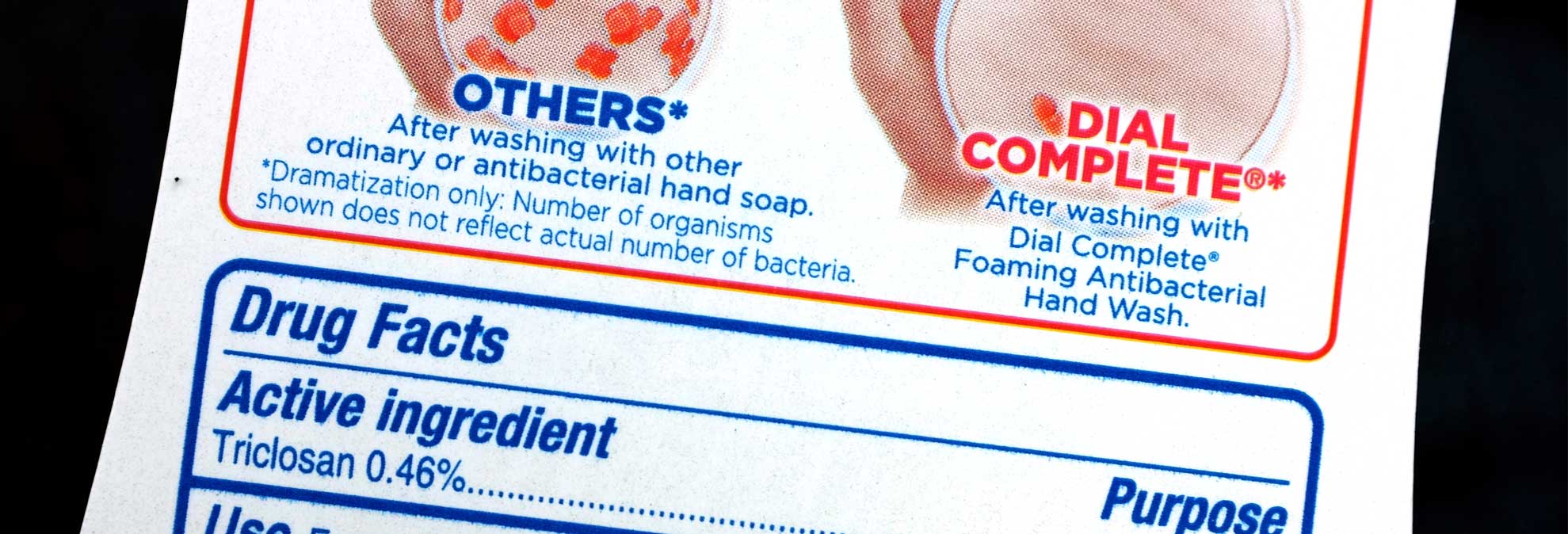
The Food and Drug Administration is banning the sale of antibacterial soaps and body washes after manufacturers failed to prove that the products’ active ingredients are safe and effective.
“Consumers may think antibacterial washes are more effective at preventing the spread of germs, but we have no scientific evidence that they are any better than plain soap and water,” said Janet Woodcock, M.D., director of the FDA’s Center for Drug Evaluation and Research. “In fact, some data suggests that antibacterial ingredients may do more harm than good over the long term.”
The ban applies to products containing 19 antibacterial ingredients, including the two most common ones: triclosan, which is found in liquid soaps, and triclocarban, found in bar soaps. Manufacturers have until Sept. 6, 2017 to either reformulate products with any of those 19 ingredients or remove them from the market.
read more at consumerreports.org